Part 1- Modeling Electron Clouds
- electron cloud is broken up into 7 layers
- the layers of the electron cloud are called either energy levels or energy shells
- on energy level 1 you can have up to 2 electrons and on energy levels 2-7 can have up to 8 electrons*
- the number of electrons in the outer layer of an atom controls how or if atoms react with other atoms
- Valence shell- outer most shell of an atom
- Valence electrons- amount of electrons you have on the outer most shell
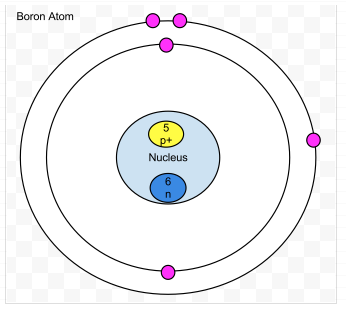
Boron-
Nitrogen
Aluminum
Phosphorus
Calcium
- Protons- 5
- Neutrons- 6
- Electrons- 5
- amount of valence- 3
- Valence shell is not full
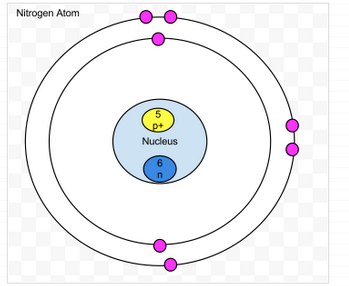
Nitrogen
- Protons-7
- Neutrons- 7
- Electrons-7
- amount of valence- 4
- Valence shell is not full
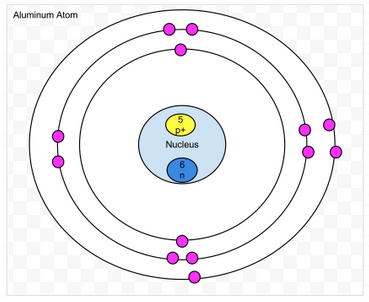
Aluminum
- Protons- 13
- Neutrons- 13
- Electrons- 13
- amount of valence- 3
- Valence shell is not full
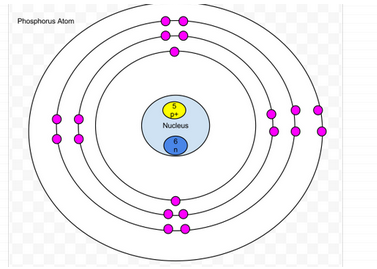
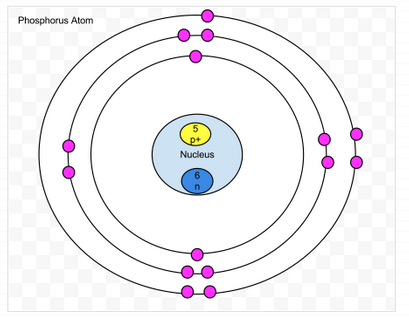
Phosphorus
- Protons- 15
- Neutrons-15
- Electrons-15
- amount of valence- 5
- Valence shell is not full
Calcium
- Protons- 20
- Neutrons- 20
- Electrons-20
- amount of valence- 2
- Valence shell is not full
Part 3- Reactivity
- Octet rule- atoms tend to want their outside energy level full of valence electrons.
- If a neutral atom's valence shell is full, then the atom will not react with other atoms.
- If a neutral atom's valence shell has space available, then the atom will react with other atoms based on the number of electrons found in the valence shell.
- Some atoms gain electrons, making the atom a negative ion.
- Some atoms tend to lose electrons and become positive ions.
- Some atoms share electrons but doesn't effect their charge.