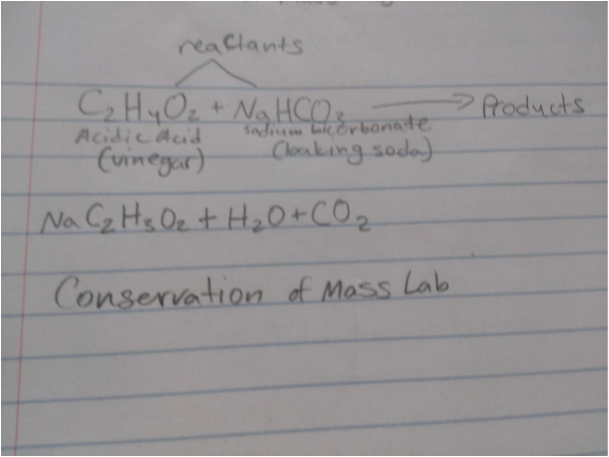Conservation of Mass Law
Mass cannot be created or destroyed.
Chemical Reaction
A Chemical reaction is when the molecular structure other than the nucleus of a substance changes. It is when two elements combine to form something new.
A substance undergoes a change to become a new substance
Indicators:
A substance undergoes a change to become a new substance
Indicators:
- A change in temperature
- Change of color
- Precipitate is formed- A solid precipitates out of a solution
- gas is given off
- Change in smell or taste
C2H4O2+NaHCO3 >>>> NaC2H3O2+H2O+CO2
Reactants Product
Sodium Bicarbonate Sodium Assetate
Reactants Product
Sodium Bicarbonate Sodium Assetate
- Gas was being produced in result of a chemical reaction when we mixed the vinegar and baking soda. The gas entered the balloon causing it to inflate. Also there was a change in temperature: it dropped.
- The final mass and the initial mass were the same.
- The vinegar was a liquid. The baking soda was a solid. The product of the two of them combined was a solid. The carbon dioxide was a gas.