Hydrogen
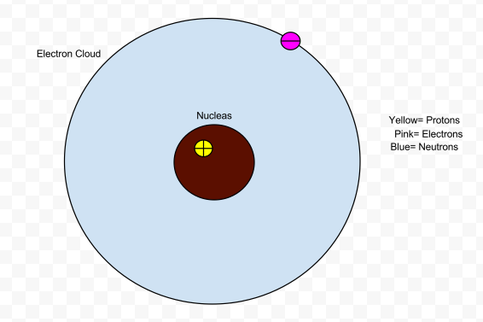
Hydrogen
Part 1
- The mass of an atom is found in the Nucleus.
- The electron cloud contributes to the volume the most.
- Nucleus has a positive charge because of the protons.
- The electron cloud is negative.
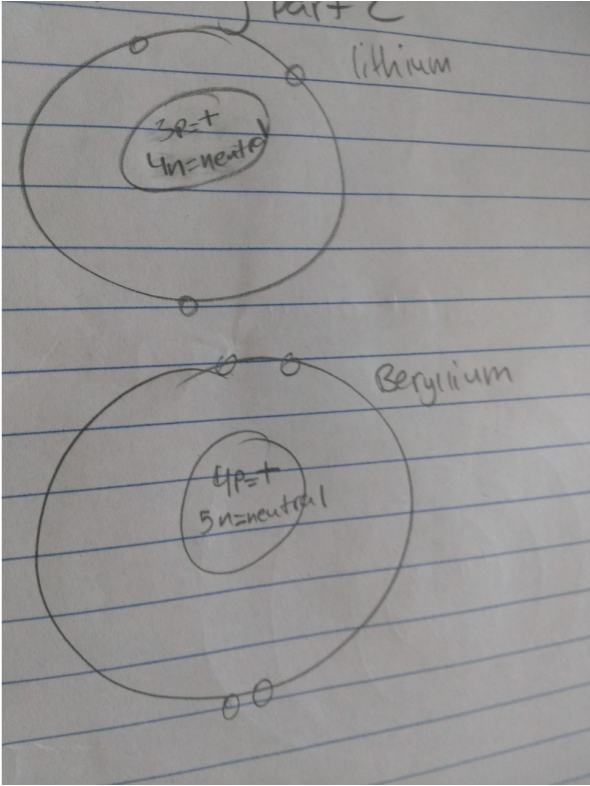
Part 2
Reflection and Conclusion
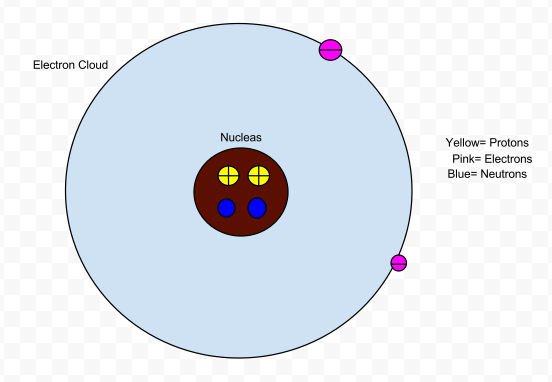
- Electrons have a negative charge and are found in the area of an atom called electron cloud.
- The atomic number in an atom is also the number of protons (mainly) or electrons in an atom.
- Protons have a positive charge and are found in the area of an atom called the nucleus.
- Neutrons do not have a charge and are found in the area of an atom called the nucleus.
- Protons and neutrons contribute mass to the atom.
- Electrons contribute to an atom's volume.
- Each proton and each neutron contribute 1 amu to an atom. An amu means Atomic Mass Unit.
- If we know the total mass of an atom in amu, we can subtract the number of electrons to calculate the number of neutrons found in the nucleus of the atom.
- In an Neutral atom the number of protons is the same as the number of electrons.
- Atomic numbers for atoms increase in increments of one for the elements on the periodic table of elements.