Comparative Lab
Problem
How much is the solution going to rise when different amounts of baking soda is mixed with a constant amount of vinegar?
Research
- When baking soda and vinegar is mixed a gas is produced.
- The temperature decreases which is called an endothermic reaction.
- Breaking the bonds between vinegar and sodium bicarbonate requires energy. Forming new bonds releases energy. Since more energy is used than released forming the product the temperature decreases.
"24 X 7." CHEMICAL REACTIONS. Fact Monster, 2013. Web. 14 Apr. 2013.
Newmark, Ann. Chemistry. New York: n.p., 2005. Print. Eyewitness Science.
Hypothesis
If I test by putting different amounts of baking soda in 3 tablespoons of vinegar then the one where I put in 4 teaspoons of baking soda is going to rise the most.
Variables
Independent: Amount of baking soda being put into each container
Dependent: The amount the solution rises
Dependent: The amount the solution rises
Materials
- 3 clear, long glasses
- 9 tbs of vinegar
- 12 tsp of baking soda
- 1 ruler
- 1 marker
- paper (to label each glass)
Procedure
- First pour 3 tablespoons of vinegar into each glass.
- Second label each glass A, B, and C.
- With the ruler measure how much liquid you're starting out with (before baking soda)
- Then start with glass A and pour 2 teaspoons of baking soda in
- Let it fizz and rise completely
- Then measure how much it rose with the ruler
- After measuring the reacted solution, subtract the reaction measurement with the initial amount
- Repeat steps 4-7 for each glass, but instead of cup A do it for cups B and C while pouring in 3tsp and 4tsp in each cup respectively
- After calculating amount risen in each cup record result in a table.
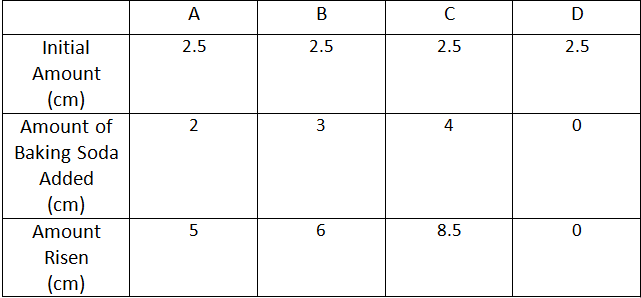
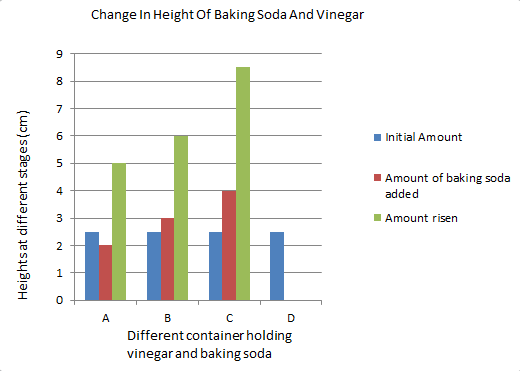
Graph/Table
The graph shows with each bar that the amount in the container is increasing. Except for the initial amount which is always the same. Each bar represents a measurement at different stages in the experiment. D is the control group.
Conclusion
My hypothesis that glass C, or the glass containing 4 teaspoons of baking soda, would rise the most is correct. The liquid inside of the glass rose more than the other 3 glasses. This is including the foam layer on top.
The data shows that when I added 3 teaspoons of baking soda to glass C, it rose way higher than the other two glasses. Glass A only got 2 teaspoons while glass B got 3 teaspoons of baking soda. The amount of vinegar that each glass started out with was kept the same. My control group was glass D. Glass D did not receive any baking soda so it didn't rise at all. When I was measuring it I made sure to measure right away when the reaction reached it's peak and measured the foam as well.
Glass C rose the most because it had the most making soda in it. The others didn't have as much baking soda mixed with the vinegar. This is because the more baking soda I mix in the more gas is formed. When there is a lot of gas and bubbles and fizz, the liquid will rise more. Like in a balloon, the more gas is blown in, the bigger the balloon gets. This is because it is taking up more space and is displacing the liquid. A chemical reaction is when the bonds in atoms are broken and new ones are created. That is exactly what happened to the baking soda and vinegar. It produces carbon dioxide, which is what humans breathe out. Some factors that may have affected the outcome was how fast I put in the baking soda. I was using a teaspoon so I had to pour it twice or three times in immediately after one another. Other than that I had no problem with this experiment. The results came out as expected.
The data shows that when I added 3 teaspoons of baking soda to glass C, it rose way higher than the other two glasses. Glass A only got 2 teaspoons while glass B got 3 teaspoons of baking soda. The amount of vinegar that each glass started out with was kept the same. My control group was glass D. Glass D did not receive any baking soda so it didn't rise at all. When I was measuring it I made sure to measure right away when the reaction reached it's peak and measured the foam as well.
Glass C rose the most because it had the most making soda in it. The others didn't have as much baking soda mixed with the vinegar. This is because the more baking soda I mix in the more gas is formed. When there is a lot of gas and bubbles and fizz, the liquid will rise more. Like in a balloon, the more gas is blown in, the bigger the balloon gets. This is because it is taking up more space and is displacing the liquid. A chemical reaction is when the bonds in atoms are broken and new ones are created. That is exactly what happened to the baking soda and vinegar. It produces carbon dioxide, which is what humans breathe out. Some factors that may have affected the outcome was how fast I put in the baking soda. I was using a teaspoon so I had to pour it twice or three times in immediately after one another. Other than that I had no problem with this experiment. The results came out as expected.