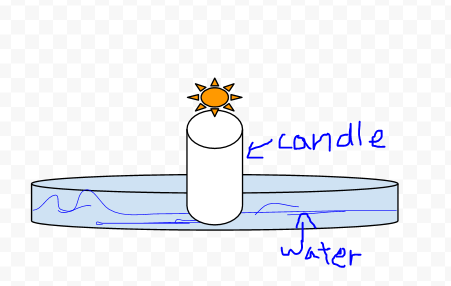
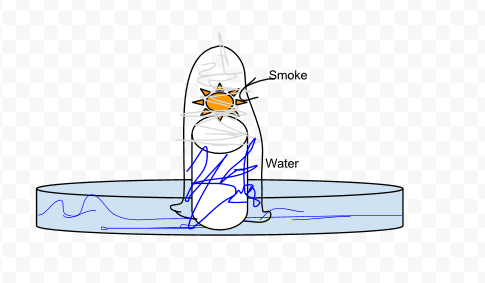
Observations
- When we put the test tube over the lit candle the flame went out.
- After the flame went out, the water started to rise up the sides of the test tube.
- The smoke was still trapped inside the test tube.
There was no oxygen left inside the test tube so it was kind of like a vacuum which sucked some water in. It was a low pressure system inside, and a high pressure system is outside.
Conclusion
- Oxygen is needed for the candle to burn.
- After we inverted the test tube, the water started to rise up inside of it. It was because there was no oxygen inside so it was like a vacuum. A low pressure system was inside the test tube. The system outside was high pressure.
- The temperature inside the test tube increased.
- It decreased.